Consumers have an expectation that the products they purchase will not cause illness or injury when used correctly. Unfortunately, this isn’t always the case. Consumers don’t generally have recourse in cases of obvious misuse or in which the customer knowingly assumes the risk, but manufacturers, retailers and other sellers can often be sued for damages caused by defective products. These claims are known as product liability cases.
For your free case evaluation, please call our experienced Alabama product liability lawyers at (251) 432-2277.
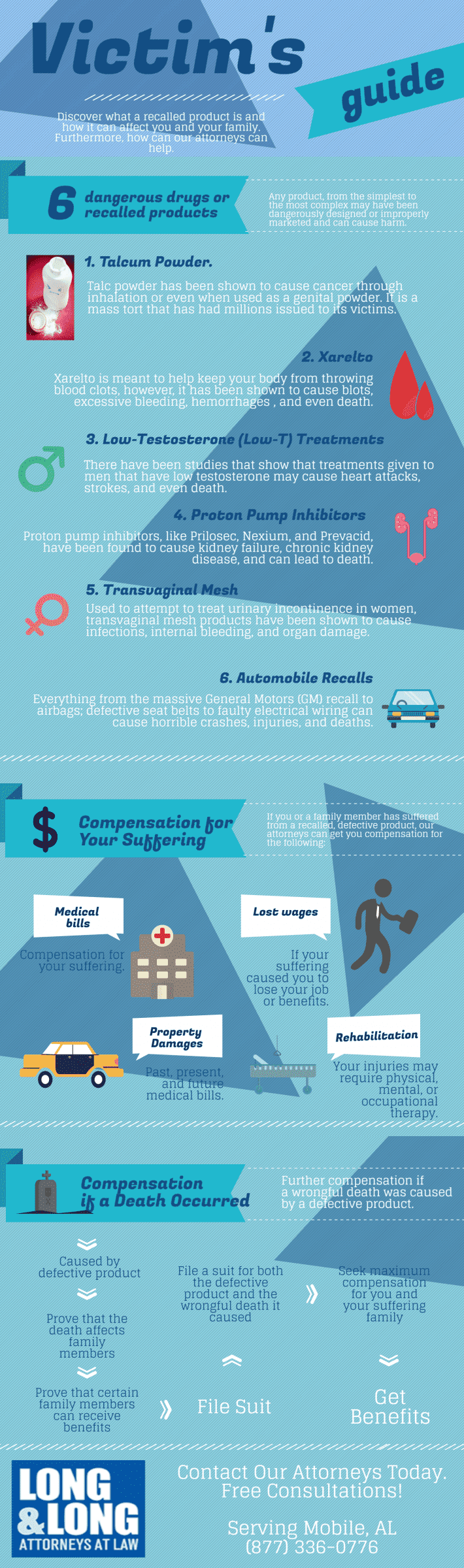
What Are Defective Products?
Any product you buy is capable of having a dangerous flaw. Defective product claims commonly arise from the following categories:
- Defective Vehicles – roof crush; airbags; seatbelt failure
- Defective Car Seats
- Smoke Detectors & Alarms
- Talcum Powder Ovarian Cancer
- Exactech® Optetrak® Claims
- Defective Medical Devices
- Dangerous Children’s Toys
- Defective Home Appliances Causing Fire
- Acetaminophen causing autism in children
-
Camp Lejeune Water
One of the most important things to remember is to preserve the evidence. For the best chance of a successful claim, the defective product should be kept in the exact condition it was in immediately after the injury occurred. The product should not be altered from this state under any circumstances.
Do I Have A Claim?
When we buy a product from a store or are prescribed a drug by a doctor, we assume the product or drug is reasonably safe for use if we use it for its intended purpose and follow any instructions. A dangerous and defective product causes injury regardless of your proper use of it.
Ready to find out if you have a claim against the maker of a defective product? If so, please call our Alabama product liability lawyers at (251) 432-2277.
Infographic: Your Guide to Defective Products
Categories of Defective Product Claims
A product defect could arise in one of three ways:
- Design defects. When a product contains a defect from the point of creation, it may be said to have a defective design.
- Manufacturing defects. These defects arise when the product is being made or assembled. Oftentimes these types of defects affect an entire lot or order, but may not affect products previously manufactured. They may also affect a single, individual item.
- Marketing defects. This is also known as failure to provide adequate warnings or instructions. It includes: improper labeling, inadequate warnings and insufficient instructions.
These are the three basic categories of product liability. After an injury, you won’t have to worry about classifying your claim. As part of our investigative process, we will determine the source of the defect and build a strategy accordingly.
In any defective product claim, the key is to demonstrate that the defect is what caused the injury. It isn’t enough to prove a defect exists. It isn’t enough to prove you were injured. You need an experienced attorney who can demonstrate exactly how the product is faulty and how that defect caused your injury.
Starting a Defective Product Claim

Types of Negligence in a Product Liability Claim
When an injured consumer attempts to recover compensation from the party responsible for the defective product, there are three types of legal theories upon which these claims are based:
- Strict product liability: This requires proving a product to be “unreasonably dangerous” because of a design or manufacturing defect. In this case, you do not necessarily have to prove that the manufacturer was aware of the defect, only that they should have been.
- Negligence: Manufacturers have an obligation to sell a product free from defects. If you can prove that the manufacturer was aware or should have been aware of their defective product, and that the product caused your injury, then you have a valid claim.
- Breach of warranty or improper labeling: Insufficient warning labels, instructions or a breached guarantee that the product was safe will need to be established. Furthermore, it will need to be established whether the defect arose in manufacture or design.
Our attorneys are extensively familiar with these details. Rely on us to develop the best possible strategy for resolving your claim.
Who Is At Fault?
Our attorneys know how to examine the history of how the product got into your hands – from design to retail sale – to determine where in the process the defect arose. Most often, the responsible party in a product liability case is one of the following:
- Manufacturers
- Designers
- Assemblers
- Installers
- Retailers
- Makers of product parts
Product manufacturers have powerful insurance companies defending them. You need equally skilled attorneys on your side. Earle W. Long and C. Bennett Long have decades of experience helping people who have suffered an injury because of the wrongdoing of another person. They have the skills and experience to properly investigate and litigate your case, and they do not back down to powerful opponents.
Common Injuries
Defective products can cause a wide variety of physical injury. Just a few examples are:
- Fractures
- Spinal injury
- Brain injury
- Burns
- Scarring and disfigurement
- Illnesses and disease
- Death
These injuries can be life-altering. Our attorneys fight aggressively to maximize your compensation for the outcomes, which may include:
- Current and ongoing medical expenses
- Hospital bills
- Prescription medication costs
- Lost income
- Pain and suffering
- Costs of rehabilitative care
We want to learn how this injury has affected your life so that we can competently and adeptly fight for every dollar you are due.
Your FREE Consultation in Alabama
At The Long Brothers, we have handled claims from across the State of Alabama. With locations in Mobile and Orange Beach, we are proud to serve people from our area. For a FREE case review, please call our Alabama product liability lawyers at (251) 432-2277. You can also contact us by completing the form on this web page.